Project Background
Reimagining How Global Regulatory Standards Are Developed
The Global Harmonization Working Party (GHWP) brings together regulatory authorities and industry stakeholders across regions to advance the harmonisation of medical device regulatory frameworks.
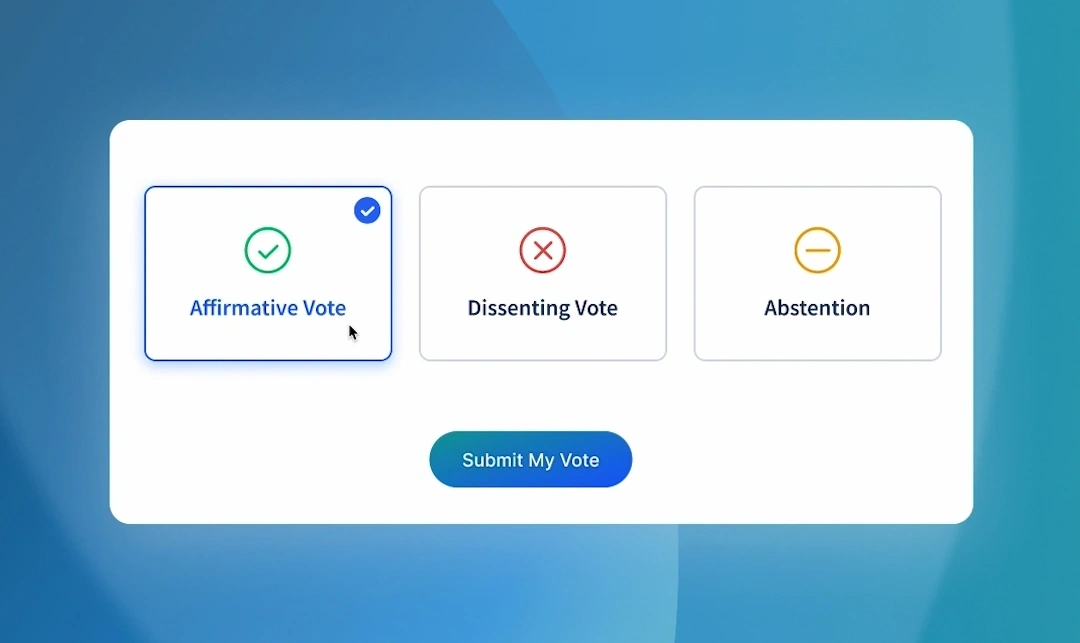
Its work depends on structured collaboration, including developing guidance documents, gathering international feedback, and formalising decisions through consensus across jurisdictions.
As participation expanded globally, the platform needed to evolve beyond a traditional website into a system capable of supporting governance, structured consultation, and formal decision-making, while continuing to serve as a trusted public resource for regulatory guidance.
Our Approach
Enabling Structured Global Collaboration
A platform approach designed to support governance, participation, and the full lifecycle of regulatory standards across international stakeholders.
Governance-Driven Platform Design
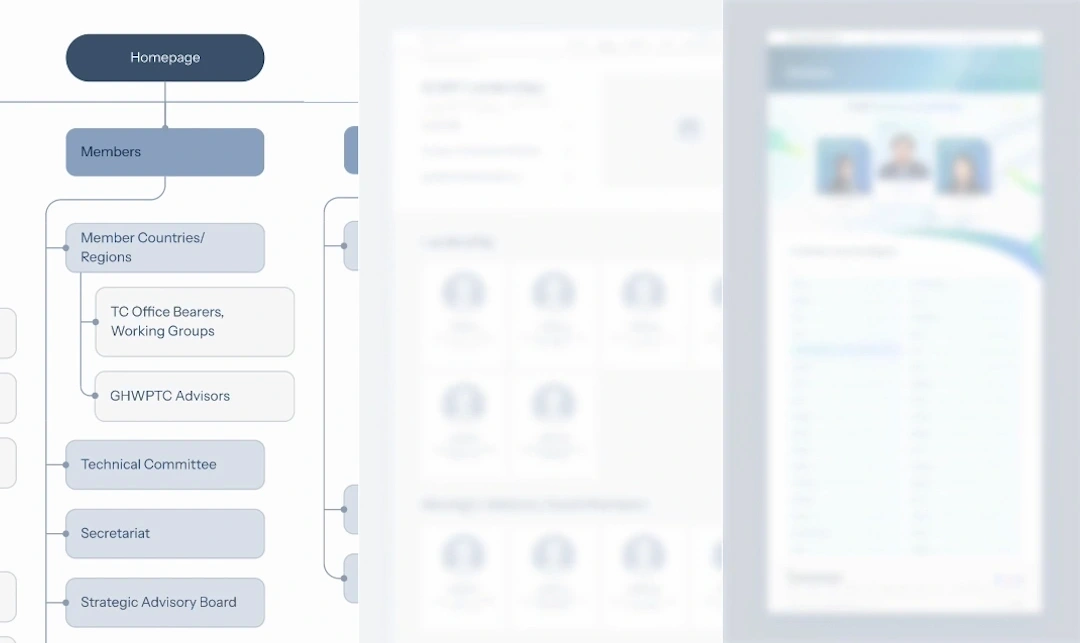
Designed the platform around structured participation, enabling authorised stakeholders to contribute, review, and make decisions within clearly defined roles and processes.
Standards Lifecycle Architecture
Transformed document management into a structured lifecycle, from drafting and consultation to final publication. Supporting how regulatory guidance is developed in practice.
Unified Public and Member Experience
Built a single platform that supports both public access to guidance and controlled collaboration among members, without fragmenting the system.
Fact & Figures
30+
Countries and Regions Participating
100+
Guidance Documents Developed and Maintained
100+
Regulatory Authorities and Industry Stakeholders